How research into bat viruses and immunity opens new doors to cancer treatment.
Bats? You mean those cave dwellers that look like a strange hybrid of rats and birds, carrying viruses transmissible to humans? Indeed, this reputation is not without reason, as bats have been the root cause for a variety of deadly diseases, including COVID-19 and SARS. However, studies at University of Rochester reveal that bats and humans possess the same antiviral genes that function differently, preventing viruses from spreading in cells (1), suggesting novel approaches to cancer treatment can be developed by researching bats.

Before understanding why bats are crucial for further research of cancer treatment, it is important to acknowledge the role of TP53. TP53 is a tumor-suppressor gene in humans that is vital for preventing tumor formation and providing protection for cells against viral infection (2). The P53 protein, encoded by TP53, inhibits the replication of COVID-19, reducing the body’s inflammatory response. However, the TP53 gene is not perfect. There is still a likelihood that viruses are able to destroy other common cellular proteins, such as MDM2 and RCHY1, which can break down P53 (5). This breakdown causes damage to the cell’s security system. This leads to the death of the cell, resulting in the virus spreading throughout the body. A similar process occurs in cancer development. As soon as P53 is threatened by other cellular proteins, the tumor starts to grow.
Bats possess a different version of the TP53 gene. Bats are known as reservoirs and act as hosts for a wide range of viruses (3). This introduces the question: how are bats immune to viruses? In 2025, researchers at the University of Rochester discovered the TP53 gene that creates the P53 protein are antivirals, and antivirals inhibit the replication of a virus (4). The only major difference is that bats have multiple copies of the same TP53 gene, which produce modified and more efficient versions of the P53 protein, leading to a better viral suppression and an anti-inflammatory response. Their version of the P53 protein can prevent viruses from entering the cell, blocking the spread of the virus (4). This is paired with continuous TP53 replication, which encodes for significantly more P53 proteins to control both viral infections and cancer cells.
In addition to the enhanced performance of P53, Alter et al’s study shows how two other factors affect the immune system of bats (1). The interferon-1 (IFN-1) molecule functions as an early-warning system, immediately activating the P53 in case of an attack from a virus (4). Unlike human cells, bats are able to produce MDM2 at a significantly higher level, reducing their susceptibility to viruses. This significantly reduces the degradation of P53.
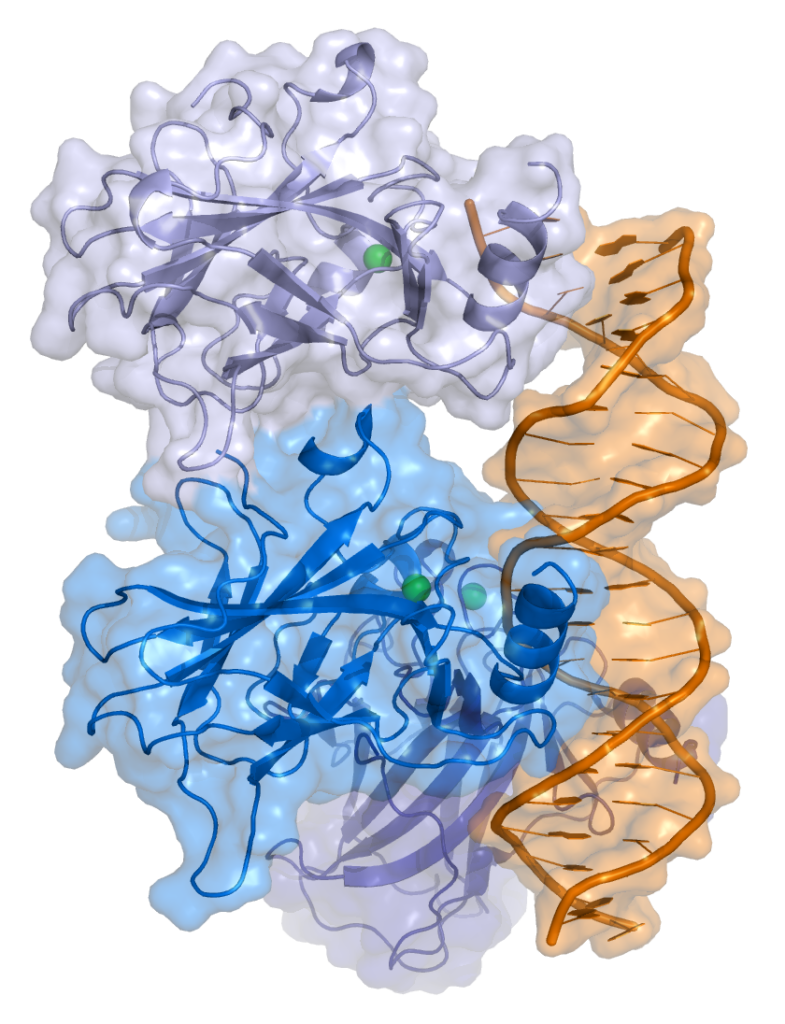
Scientists are making connections of P53 in human cells and bat cells and trying to enhance the functions of P53 in human cells, and this experiment offered a new perspective for cancer research. However, a major difference between human cells and bat cells is that bats are able to withstand a dramatic increase in P53 proteins without harm to the cell. For humans, an increase in P53 proteins causes irreversible bodily damage. In bats, an enzyme called telomerase is deployed. Telomerase compensates damage done by the P53 when it fights cancer. Also because of this challenge, scientists are currently researching the combined effects of regenerating enzymes telomerase and P53 by ensuring that production of telomerase can be increased safely, as an excessive amount of telomerase causes cells to divide uncontrollably. So while telomerase has its perks, it also may cause more cancerous cells to be replicated (4).
As scientists explore new options for cancer treatment, this discovery can help fill conceptual gaps in previous research. While it is important to take caution against viral spills from bats, it is equally important to remove the bias against them, as they can contribute to groundbreaking scientific research.
Images
- https://kids.nationalgeographic.com/animals/mammals/facts/vampire-bat
- https://en.wikipedia.org/wiki/P53
Bibliography
- Athar, F., Zheng, Z., Riquier, S., Zacher, M., Lu, J. Y., Zhao, Y., … & Gorbunova, V. (2025). Limited cell-autonomous anticancer mechanisms in long-lived bats. Nature Communications, 16(1), 4125.
- Center, N. (2001). Genes and Disease. Nih.gov; National Center for Biotechnology Information (US). https://www.ncbi.nlm.nih.gov/books/NBK22183/
- Mackenzie, J. S., Childs, J. E., Field, H. E., Wang, L. F., & Breed, A. C. (2016). The role of bats as reservoir hosts of emerging neuroviruses. In Neurotropic Viral Infections: Volume 2: Neurotropic Retroviruses, DNA Viruses, Immunity and Transmission (pp. 403-454). Cham: Springer International Publishing.
- Orr, L. (2025, June 10). Why don’t bats get cancer? News Center. https://www.rochester.edu/newscenter/why-dont-bats-get-cancer-655922/
- Wang, X., Liu, Y., Li, K., & Hao, Z. (2023). Roles of p53-mediated host–virus interaction in coronavirus infection. International journal of molecular sciences, 24(7), 6371.





Comments are closed.