The mechanics and accessibility of testing techniques in the US.
On March 10, 2020, the medical staff, faculty and trainees of the University of Washington Medical Center created one of the first drive-through testing stations in the country, right inside of their parking lot. Within days, hundreds of cars full of anxious patients were lined up outside its gates, waiting to get tested for the novel coronavirus, which causes the respiratory disease COVID-19. As the novel coronavirus has rapidly spread throughout the United States, diagnostic testing has become more crucial than ever in halting the spread of the virus and protecting the vulnerable. Drive-though clinics like those in Seattle have multiplied as the US government scrambles to make testing available across the nation.
How does coronavirus testing work?
Two main techniques are currently used to test for the coronavirus: molecular and serological.
Molecular testing, also known as PTC testing, was the first technique to be deemed effective by the Center of Disease Control and Prevention (CDC) and the World Health Organization (WHO). This test analyzes a sample collected from the patient in search of genetic material from SARS-CoV-2, the virus that causes COVID-19 (1). Each specimen is collected by inserting a six-inch swab into the back of a nasal passage, where SARS-CoV-2 is usually found, and rotating the swab for fifteen seconds. The sample is then sent to laboratories for analysis.
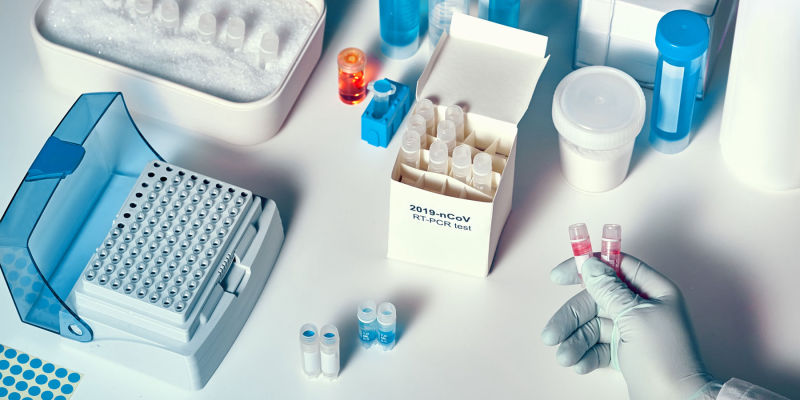
The technique used to examine this data is called reverse transcription polymerase chain reaction (RT-PCR). In RT-PCR, the genetic material in the sample is replicated and amplified in order to compare its information with that of the coronavirus sequence. This process can take anywhere from fifteen minutes to two hours, depending on the machinery used. Currently, the Federal Drug Administration (FDA) has approved more than 25 manufacturers to conduct molecular tests (1).
Serological testing is the second method of testing for COVID-19. Unlike molecular testing, it measures the amount of antibodies or proteins present in the blood when the body is responding to a specific infection (1). There are two main types of antibodies which signal the presence of the virus: immunoglobulin M (IgM) and immunoglobulin G (IgG). The presence of IgM indicates recent infection, while IgG antibodies indicate that a patient is in a later stage of the infection.
What do the results mean?
If a patient tests positive using a molecular diagnostic test, they have an active COVID-19 infection and must follow CDC guidelines on how to properly isolate and remain healthy. If the results are negative, there was likely no infection on the sample collected.
However, low levels of infection may still be present in the body even if a patient tests negative. This can occur when the specimen does not include enough genetic material to provide accurate results, or when chemicals added in the laboratory distort the results (8). Data collection thus far has shown that the chances of false positives are very rare, however, false negatives occur 30% of the time.
The same cannot be said about serological tests. According to initial studies, this form of testing is less dependable; at least 42% of the tests are incorrect (8)—this includes both false positive and false negative outcomes. If a serological test is positive, this means that the patient may either have an active infection or that they have just defeated the virus and developed immunity. As a result, doctors advise all their patients getting tested to behave as if they have the virus, regardless of the results, in order to limit its possible spread.
How accessible are the tests in the United States?
Despite considerable advancements in the accuracy of diagnosing methods, the US is facing extreme test kit shortages due to high demand. This is reflected in the number of tests the US carries out each day. Currently only 320 out of 100,000 people are tested each day, while in countries such Italy and South Korea, roughly 800 of every 100,000 people have been tested for the virus (1, 2). This has left millions of high risk medical workers, and patients demonstrating symptoms unable to receive a test.
Furthermore, many argue that this situation has also sparked the conflict of social inequities and preferential treatment. Throughout the past month, asymptomatic politicians, celebrities and athletes, have obtained immediate access to extremely limited tests and exclusive treatment, solely because of their status.
The clearest example is that of Heidi Klum, a social media influencer and actress, who used Instagram as a platform to express her anger with the inability to receive a test by two of her physicians, regardless of not passing the required CDC guidelines. In less than 24 hours, her account was flooded with clinics offering free tests; within days she announced that her test results were negative for the Covid-19 virus.
However, celebrities are not the only ones publicly using their connections in order to get tested. Close allies of President Trump, such as Senator Lindsey Graham, Republican of South Carolina, were also able to immediately obtain tests (4).
This prioritization of the rich and famous has only hurt the overall health of the nation. Unable to receive tests, people in need are more prone to spreading the virus to their living facilities without receiving treatment. This will cause a larger outbreak in those areas and limit the ability of their community to deal with the outbreak. The struggle to get tested has sparked the circulation of the tweet #CDCWontTestMe, throughout the past month.
Nevertheless, the federal government has made an effort to increase COVID-19 testing availability in all states. Furthermore, it has passed several stimulus packages to support families in financial strain.
On March 18, the federal government enacted the Families First Coronavirus Response Act, which ensures that COVID-19 testing is free for everyone, regardless of their insurance status or provider. Nevertheless, patients diagnosed with this virus who require hospitalization or additional assistance may be responsible for added costs (1). On March 27, Congress passed the Coronavirus Aid, Relief, and Economic Security (CARES) Act which contributes to the funding for hospitals, testing facilities, emergency supplies, aiding drugs, private protection equipment, and device shortages, such as ventilators, across the US (1).
Who is eligible to be tested?
As a result of accessibility limitations, the CDC and local health department has issued guidelines on who can receive a test. In Massachusetts, all who demonstrate symptoms and have been approved by their primary physician are permitted to be tested. Most tests are performed in areas that have high case levels, such as New York and California.
How accessible are tests in Third World Countries?
Regardless of massive test shortages across the country, the US is facing one of the best case scenarios during this pandemic. Dozens of third world countries throughout Africa and Latin America have been left on the brink of destruction. Recent studies have shown that many of these countries have an underfunded, underequipped and fragile health system which is only able to provide one properly equipped intensive care bed per million residents.
This situation is only worsened with the severe shortage of testing (7). Scientists in these areas have been told that their orders for testing kits could not be filled for months to come. This is because the majority of test kits and essential Personal Protective Equipment (PPE), such as masks, have been stripped away by powerful countries such as the US and Germany which have demanded first-in line access to vital supplies. Furthermore, with the steep increase in kit prices, third world countries could not afford access to large quantities of tests for a considerable period of time. As a result, they are left to the mercy of non-profit companies such as UNICEF to help them get the supplies they need. Scientists throughout the world believe that without testing, these countries remain unprepared for the threat COVID-19 poses (8).
The coronavirus pandemic has shone light on the underlying problems in our society. From the government’s insufficient preparation, to class inequality, this challenging time has sparked conflict and led to disaster. Hopefully, in times like these, we can improve socioeconomic divides for a more equitable society.
– Andri Kadaifciu
References
- Billingsley, A. (2020, April 9). Coronavirus (COVID-19) Testing Types, Methods, Efficacy, and Availability – GoodRx. Retrieved from https://www.goodrx.com/blog/coronavirus-covid-19-testing-updates-methods-cost-availability/#molecular-test
- An antibody test for the novel coronavirus will soon be available. (n.d.). Retrieved from https://www.economist.com/science-and-technology/2020/04/02/an-antibody-test-for-the-novel-coronavirus-will-soon-be-available
- Top Medical Testing Kits Suppliers in the US (including Coronavirus/COVID-19 Tests). (n.d.). Retrieved from https://www.thomasnet.com/articles/top-suppliers/medical-testing-kits-suppliers/#_Resources_for_Covid-19
- Schaffer, J. (2020, March 21). Why are the rich and famous getting coronavirus tests while we aren’t? | Jennifer Schaffer. Retrieved from https://www.theguardian.com/commentisfree/2020/mar/21/rich-famous-coronavirus-tests-covid-19-tom-hanks
- Twohey, M., Eder, S., & Stein, M. (2020, March 19). Need a Coronavirus Test? Being Rich and Famous May Help. Retrieved from https://www.nytimes.com/2020/03/18/us/coronavirus-testing-elite.html
- Alexis C. Madrigal, R. M. (2020, April 1). Private Labs Are Fueling a New Coronavirus Testing Crisis. Retrieved from https://www.theatlantic.com/health/archive/2020/03/next-covid-19-testing-crisis/609193/
- Bradley, J. (2020, April 9). In Scramble for Coronavirus Supplies, Rich Countries Push Poor Aside. Retrieved from https://www.nytimes.com/2020/04/09/world/coronavirus-equipment-rich-poor.html
- Lanese, N. (2020, April 3). Even if you test negative for COVID-19, assume you have it, experts say. Retrieved from https://www.livescience.com/covid19-coronavirus-tests-false-negatives.html
Images





Be First to Comment