How tiny molecules speed up vaccine production.
Despite its microscopic size, SARS-Cov-2, the virus responsible for the disease COVID-19, continues to ravage the world. The molecules of COVID-19 vaccines, designed to counter the virus, will arrive in an almost equally small package: lipid nanoparticles (LNPs).
The vaccines made by Pfizer-BioNTech and Moderna both use messenger RNA (mRNA) in their vaccines. mRNA acts like a photocopy of a DNA segment and is used by ribosomes––located inside the cell––to make proteins. Pfizer and Moderna’s vaccines use mRNA to reproduce the spike proteins found on the surface of SARS-Cov-2, which the virus uses to enter cells. The ones produced by the body are then presented to the body’s immune system so that it learns to recognize the virus. Once the mRNA has been used by the ribosome, it is quickly destroyed by RNAse, an enzyme that breaks down free floating RNA in the body.
Although an mRNA vaccine has never been used before due to fragility problems, the concept of one has existed for many years. The antigens required to create mRNA vaccines are much easier to produce than other types of vaccines: scientists only have to copy and reproduce part of the virus’ genome, easily done by using a polymerase chain reaction (PCR). However, the rapid degradation of RNA inside the body makes delivering mRNA to the inside of a cell one of the hardest challenges to overcome in the creation of a successful vaccine; only recently have scientists realized that lipid nanoparticles are a suitable carrier for mRNA.
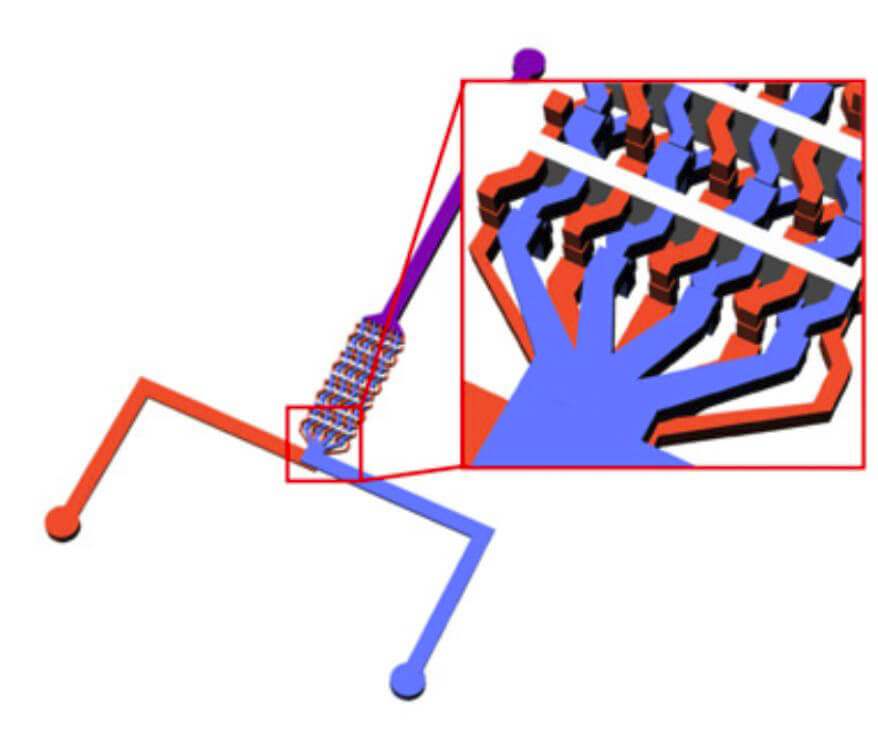
Moderna and Pfizer both encapsulate mRNA inside LNPs. They create such particles by running a solution containing the lipids that constitute the LNP alongside another solution that contains the mRNA in a buffer, such as acetic acid, in a microfluidic mixer. Because of the close proximity of these two solutions, they will spontaneously and rapidly combine to produce lipid nanoparticles that contain mRNA. Once inside a nanoparticle, the mRNA can evade destruction from RNAse. Eventually, the nanoparticle is absorbed into a cell through endocytosis, and pH-sensitive lipids in the nanoparticle will allow it to release the mRNA into the cell. While Moderna and Pfizer have not released the exact lipids and ratios of lipids in their vaccines to the public, a typical LNP is composed of four different types of lipid: an ionizable lipid, a phospholipid, a cholesterol, and a PEG-lipid. LNPs and other nanomaterials can also use the lymphatic system to traverse the body. This can be especially useful for transporting LNPs to lymph nodes near the lungs, where they can join the front lines of the fight against respiratory diseases.
Moreover, LNPs can provide synchronous delivery of mRNA and adjuvants, molecules that stimulate the immune system and boost immune response to the antigens in all vaccines. Many adjuvants have failed clinical trials due to toxicity issues, since adjuvant molecules can end up in cells without mRNA and stimulate an unwanted immune response. Though, synchronous delivery can solve that problem. By reducing off-target delivery, LNPs reduce the necessary dosage of mRNA needed to induce immunity; doses with less mRNA and more effective adjuvants are as productive as a greater dosage of mRNA since the immune response will be more focused.
By effectively delivering mRNA to cells, liquid nanoparticles have accelerated vaccine production for the fight against contagious diseases. It took the world a year to come up with a COVID-19 vaccine, but the delays were only due to necessary caution surrounding new lipid nanoparticle technology. Now that lipid nanoparticles have proved themselves effective, the world could regularly see vaccines for new diseases in less than a year after discovery-a drastic improvement from the previous record held the mumps vaccine which took 4 years to create. Although invisible to the naked eye, they have already saved millions of lives.
– Timothy Guan
Image Sources
- https://www.elveflow.com/microfluidic-reviews/microfluidic-flow-control/microfluidic-mixers-a-short-review/
- https://www.frontiersin.org/articles/10.3389/fchem.2020.589959/full




Comments are closed.