The discovery of saccharin and its lasting impact on food science
In 1879, chemist Constantin Fahlberg sat down for dinner and noticed something strange: his food tasted unusually sweet. Fahlberg had been working alongside another chemist, Ira Remsen, at Johns Hopkins University, investigating coal tar derivatives and the oxidation of o-toluenesulfonamide, a compound used in chemical research and industrial processes (1). Curious about the unexpected sweetness after touching his food, Fahlberg returned to the lab and, together with Remsen, discovered that the source was a compound formed during his experiments, later known as saccharin (2).
Saccharin is an artificial, non-nutritive sweetener—meaning it provides no nourishment or significant caloric value—and an organic compound that exists either as an insoluble solid or in more soluble salt forms, which are created when saccharin reacts with metals such as sodium or calcium. In these salt forms, the chemical structure of saccharin stays the same, while only its physical properties, such as solubility, are altered. Estimated to be 200 to 700 times sweeter than sucrose, saccharin is often described as having a slightly bitter or metallic aftertaste. Chemically, saccharin is a white crystalline solid in its insoluble form and melts at temperatures between 228.8 and 229.7 degrees Celsius. In its salt forms, it appears as a white crystalline powder, a property that makes it easier to dissolve and incorporate into foods. Saccharin is soluble in water and remains stable across a pH range of 2 to 7 and temperatures up to 150 degrees Celsius (1).
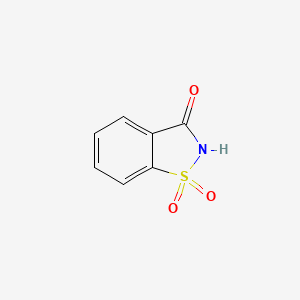
Following the chemical’s discovery, Fahlberg and Remsen developed a method of synthesizing saccharin from o-sulfamoylbenzoic acid, allowing for its production beyond the laboratory and at an industrial scale (2). Saccharin and its salts do not occur naturally, meaning they can only be produced synthetically rather than through agricultural processes (3). Because saccharin is not metabolized by the human body and is excreted unchanged, it provides sweetness without adding calories or contributing to tooth decay (1).
Despite its usefulness, saccharin quickly became a controversial substance. In 1906, public health concern over food additives intensified following the publication of Upton Sinclair’s The Jungle, a novel that exposed the unsanitary conditions in the American meatpacking industry. This prompted U.S. Department of Agriculture chemist Harvey Wiley to propose banning saccharin; however, President Theodore Roosevelt rejected the proposal, dismissing the claims of harm and ultimately firing Wiley. This decision may have been influenced by the fact that Roosevelt himself used saccharin to lose weight (2). Saccharin’s popularity surged during World War I and later World War II due to sugar shortages, and by the 1960s, it had become a major component of the rapidly growing diet food industry under brand names such as Sweet’n Low (2, 4).

As saccharin became more widely adopted, its unique properties made it the first widely used non-nutritive sweetener and rendered it particularly beneficial for individuals with diabetes or others seeking to avoid sugar (2). Its applications later expanded to diet soft drinks and other low-calorie foods. It is currently approved for use as a food additive in most countries, including the United States (1). Beyond food and beverages, saccharin has also been used as a nickel electroplating brightener that improves the appearance of metal coatings, an animal feed sweetener, and an anaerobic adhesive accelerator, which speeds the curing of some industrial glues (3).
Further concerns arose in the 1970s when studies linking saccharin to bladder cancer in rats led Congress to pass the Saccharin Study and Labeling Act in 1977, requiring cancer warning labels on products containing saccharin (5). Subsequent research demonstrated that the cancer mechanism observed in rodents did not apply to humans, and in 2000, the mandatory warning labels were removed (4).
Saccharin’s accidental discovery has influenced how artificial sweeteners are evaluated, from scientific legitimacy to the regulatory scrutiny that often accompanies them. If one overlooked compound in a coal tar laboratory could open that door, who knows what other possibilities remain hidden in the byproducts of modern scientific research?
Bibliography
- Saccharin | chemical compound | Britannica. (2019). In Encyclopædia Britannica. https://www.britannica.com/science/saccharin
- American Chemical Society. (2019, July 1). Saccharin. American Chemical Society. https://www.acs.org/molecule-of-the-week/archive/s/saccharin.html
- Humans, I. W. G. on the E. of C. R. to. (1999). Saccharin and its salts. Www.ncbi.nlm.nih.gov; International Agency for Research on Cancer. https://www.ncbi.nlm.nih.gov/books/NBK402047/
- Saccharin | EBSCO. (2022). EBSCO Information Services, Inc. | Www.ebsco.com. https://www.ebsco.com/research-starters/history/saccharin
- FDA. (2023). Milestones in U.S. Food and Drug Law. FDA. https://www.fda.gov/about-fda/fda-history/milestones-us-food-and-drug-law
Images




Comments are closed.