Telomere-to-Telomere consortium fully sequenced the human genome with never before studied sections of our DNA.
“Knowing yourself is the beginning of all wisdom.”
Aristotle
Understanding what makes us human on a genetic level is one of science’s greatest goals. Recently, geneticists have inched a step closer by completing a map of the entire human genome. While this revolutionary achievement will help to show the influence of genes on an individual’s psychology, it was almost certainly not what Aristotle meant by “knowing yourself.” Knowledge about our genetic foundation helps scientists understand why we have certain traits and may offer innovative therapies for genetic diseases (5).
In 2003, the Human Genome Project––funded with $3 billion––announced that the entire human genome had been sequenced (3). In reality, 8% of the human genome still remained unsequenced. While 8% doesn’t seem like a substantial part of our genome, it contains around 200 million DNA bases and includes entire parts of the chromosome such as the centromeres and telomeres (5). The Human Genome Project prioritized the sequencing of euchromatin, as at the time, technology was not advanced enough to sequence any other regions (3). It was also mapped first because euchromatin codes for proteins, which were thought to be the most important part of the genome. The euchromatin generally contains DNA in loose packages, which makes it the easiest region to sequence (3). The geneticists at the Human Genome Project did not sequence the heterochromatin, which is tightly wound, usually including the far ends and middle parts of the chromosome (5). Heterochromatin contains many repeated sequences, and prior it was thought to be mostly unimportant (3). This changed when researchers from the Telomere-to-Telomere (T2T) consortium discovered that this 8% of unsequenced DNA is important to cell division, organization of the genome, and controlling which genes are expressed (3).
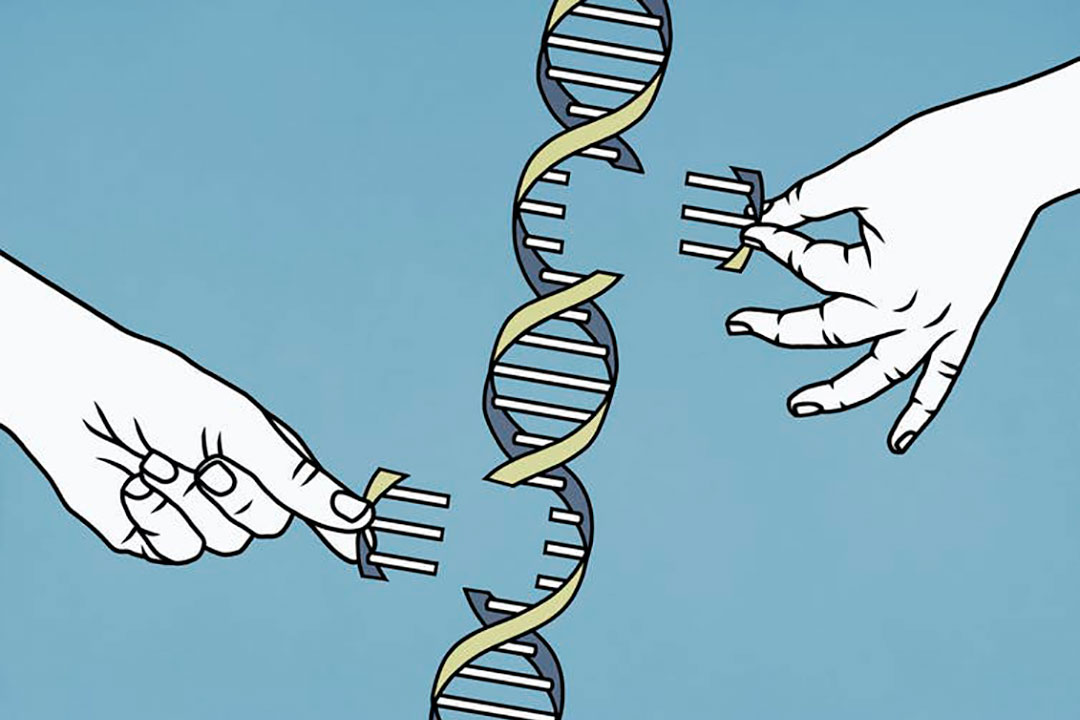
T2T consortium, the flagship study in the investigation of our genome, is made up of researchers at National Institute of Health (NIH) and other various academic institutions (5). T2T consortium takes its name from telomeres, which are the repetitive pieces of DNA at the end of chromosomes (4). It is important to note that “there is no such thing as the human genome” (5). Two people can have large portions of their genome that are very similar if not identical and smaller parts that are substantially different. The genome that T2T consortium sequenced was not actually from a person. Instead, the scientists sequenced a hydatidiform mole, which is a tumor that emerges when an empty egg is fertilized by a sperm (5). These tumors were used by the researchers because they only contain the paternal chromosomes. People generally have two genomes in each cell, one from their mother and the other from their father. Attempting to sequence a full cell with both chromosomes would make it difficult to sort between the two (5). The T2T consortium study combined highly accurate long read sequencing technology from Pacific Biosciences’ “HiFi” circular consensus sequencing with Oxford Nanopore Technology (ONT) sequencing to resolve breaks and inconsistencies in the genetic code (4). The team then combined the data from these different techniques to fit base pair sequences together, as one might do with a jigsaw puzzle. Thus, 20 years after the Human Genome Project ended, the first complete human genome has been sequenced.
The implications of the T2T consortium’s study are still not fully understood, but it is clear the effect they will have on upcoming therapies. I was able to conduct an interview with Mike Vincent, the Chief Scientific Officer of Inflammation and Immunology at Pfizer, to get his perspective on the impact of the study. While there were a relatively small number of new proteins identified, they each were crucial for creating new therapies.

“Probably most impactful is a better understanding of the center and ends of chromosomes, which could have implications for therapies targeting cancer and aging,” Vincent said. The sequencing of centromeres, the connection between the sister chromatids in a chromosome, gives researchers a better understanding of cell division, especially as we age. Many scientists believe that it is errors in cell division which cause age related cancers. The discoveries made by T2T are revolutionary, but Vincent explained that revelations about our genetic code are just beginning.
“It’s tempting to think that now that we have the sequence, we know everything we need to know. Instead, it’s really the beginning of generating insights. It’s a little like having a map, which is something very different than exploring terrain,” Vincent said. The understanding of how to map someone’s genes could lead to personalized therapies. There are already genetic therapies being produced, such as T-Vec, which is a treatment for melanoma. The treatment uses a modified virus to kill cancer and attract immune cells (6). It is an exciting time, as we cast an eye to future possibilities knowing the details of our genetic code.
- Lumb, D. (2022, April 1). Scientists Finally Sequence the Entire Human Genome. CNET. https://www.cnet.com/science/biology/scientists-finally-fully-sequence-the-human-genome/
- Jarvis, E. (2022, March 31). The human genome is, at long last, complete. The Rockefeller University. https://www.rockefeller.edu/news/32087-the-human-genome-is-at-long-last-complete/
- Green, H. (2022, April 8). We Hadn’t Sequenced the Human Genome…Until Now | SciShow News. YouTube. https://www.youtube.com/watch?v=cneFGjEL9D0
- Wrighton, K. (2021, February 10). Filling in the gaps telomere to telomere. Nature Research. https://doi.org/10.1038/d42859-020-00117-1 (4)
- Saey, T. (2022, March 31). We finally have a fully complete human genome. Science News. https://www.sciencenews.org/article/human-genome-complete-dna-genetics (2)
- Gene Therapy Successes. (2012). Utah.edu. (6) https://learn.genetics.utah.edu/content/genetherapy/success/
Images:
- https://www.nature.com/articles/d41586-021-01506-w
- https://www.gavi.org/vaccineswork/human-genome-project-pieced-together-only-92-dna-now-scientists-have-finally-filled




Comments are closed.